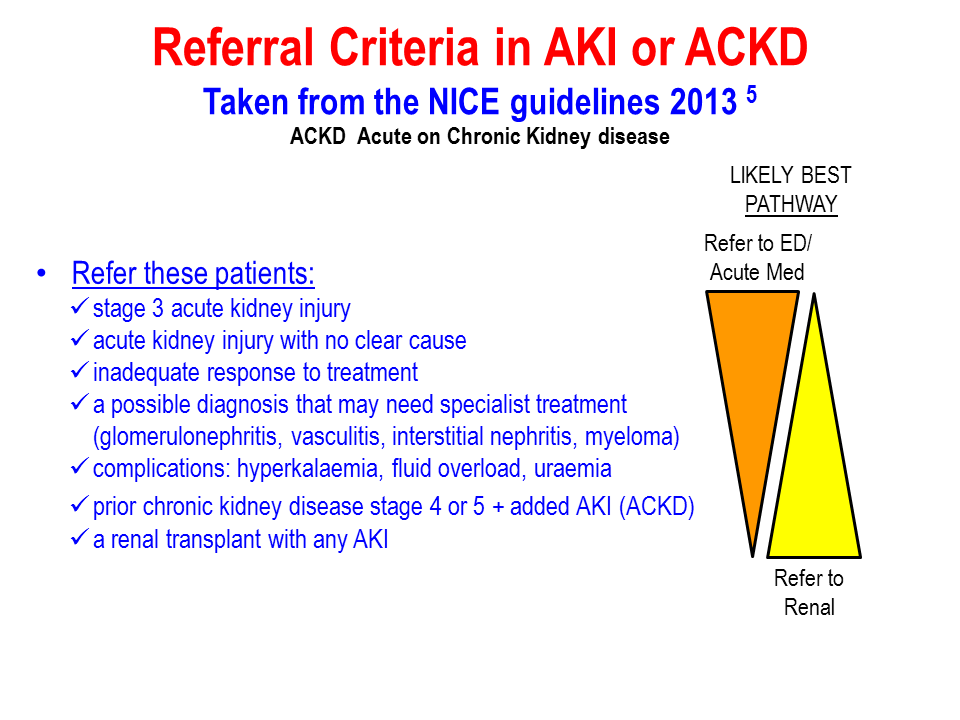
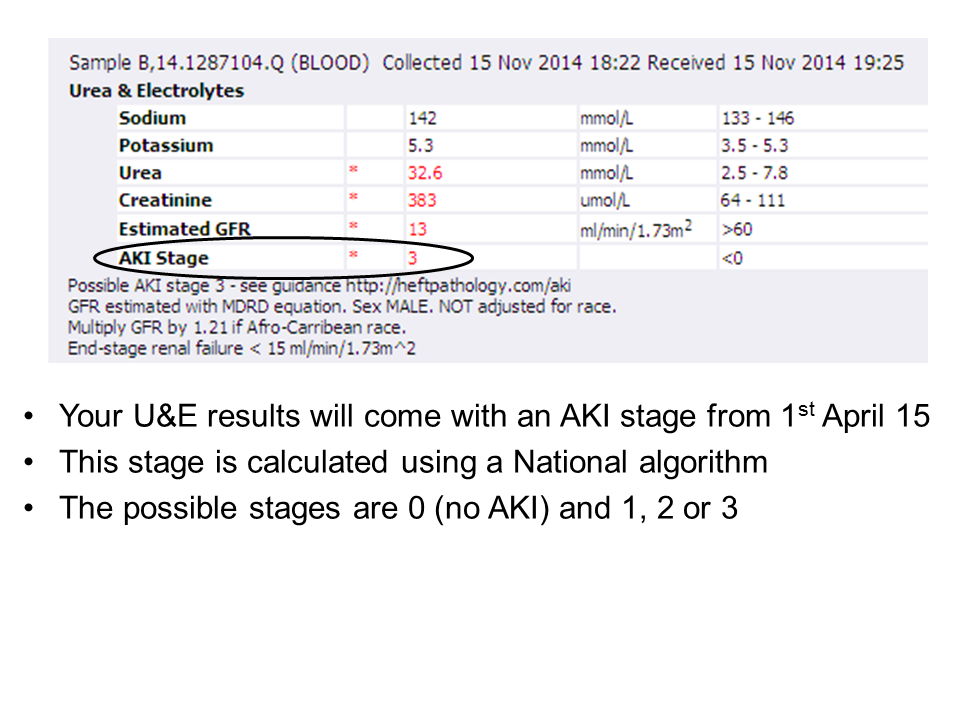
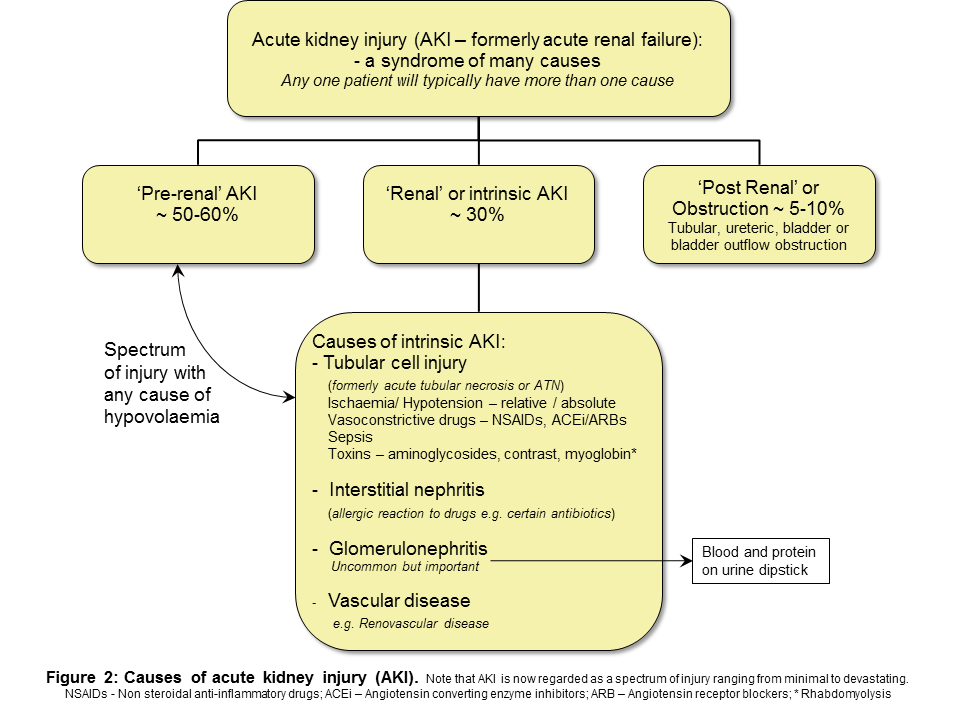
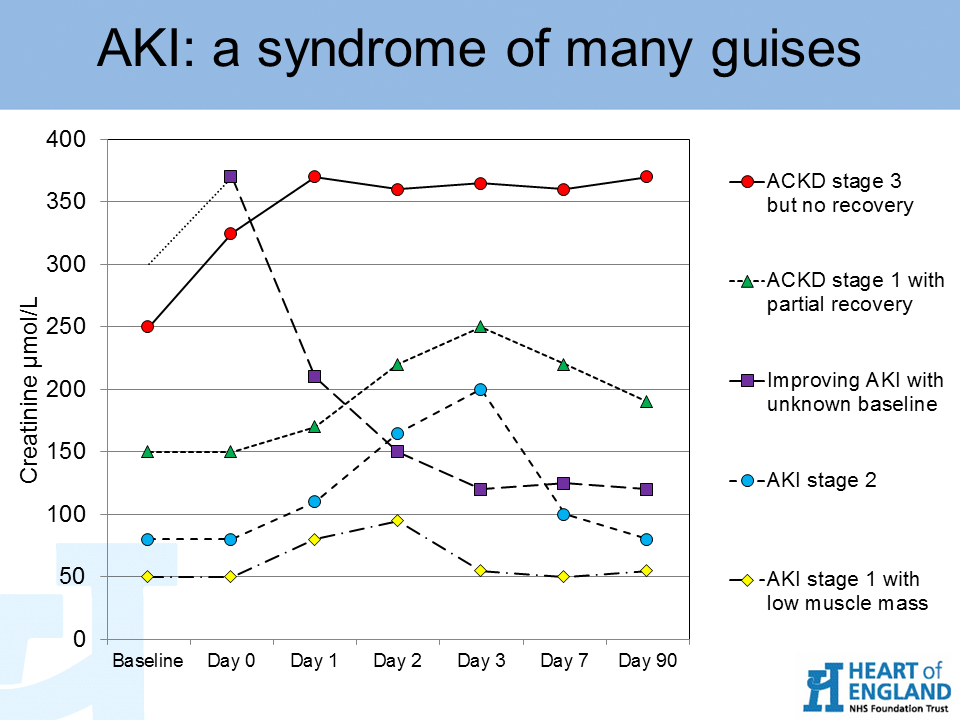
Tumour Marker Use in Primary Care
In the last 5 years requests for tumour marker tests from Primary Care have more than doubled. This high use in Primary Care is worrying because the majority of tumour markers (eg. CEA, CA19-9) are neither specific nor sensitive enough for use in the diagnosis of malignancy. See this link for a summary of the main tumour markers, their uses and limitations.
The main use for tumour markers is in monitoring disease progression, treatment or recurrence of a histologically diagnosed cancer. A recent audit of Primary Care requests for tumour markers found that only 9% of CEA and 4% of CA19-9 were requested for these reasons; the rest being for non-specific symptoms.
In contrast to the above, CA125 and PSA do have use in diagnosis of their related cancers, however it should also be noted that these are still only a diagnostic aid and should be used with caution as both can be raised in a number of other benign conditions (see table). Please click the relevant links below of links to guidelines relating to their use in Primary Care.
CA125 link https://pathways.nice.org.uk/pathways/ovarian-cancer
PSA link https://www.gov.uk/guidance/prostate-cancer-risk-management-programme-overview
For symptoms and referral guidelines of other malignancies see the NICE Suspected Cancer Recognition and Referral guidelines. http://pathways.nice.org.uk/pathways/suspected-cancer-recognition-and-referral
You can also use the search bar or test database on this website to find more specific information on the use of each tumour marker.
|
Tumour marker |
Main application |
Other tumour elevations |
Other limitations |
|
CEA |
Monitoring colorectal adenocarcinomas |
Breast, lung, gastric, mesotheliomas, oesophageal and pancreatic |
Raised in smokers Raised in other benign renal, liver, lung or GI disease Poor sensitivity in early disease and may be absent/low in poorly differentiated tumours |
|
CA19-9 |
Monitoring pancreatic carcinoma |
Raised in obstructive jaundice, cholestasis, cirrhosis, pancreatic hepatitis and non-malignant GI disease. Not present in those negative for the Lewis blood group determinant. |
|
|
CA125 |
Monitoring ovarian carcinoma |
Raised in patients with ascites, pleural effusions or free fluid in the pelvis Raised in patients with congestive heart failure Raised in benign renal and liver disease and other adenocarcinomas Mildly raised in menstruation and the first two trimesters of pregnancy Can be raised in endometriosis |
|
|
CA15-3 |
Monitoring breast cancer |
Lung, colon, ovary |
Raised in benign liver, breast, ovarian disease |
|
AFP |
Diagnosis and monitoring of hepatocellular carcinoma and germ cell tumours |
Gastric and other GI (oesophageal, pancreatic) |
Raised in pregnancy and neonates Raised in benign liver disease |
|
PSA |
Diagnosis and monitoring of prostate carcinoma |
Also elevated in benign prostatic conditions Increases with age (as prostate size increases) Elevated in UTI, catheterisation, prostatitis or other prostate manipulation |
|
|
hCG |
Diagnosis and monitoring of germ cell tumours and gestational trophoblastic neoplasia |
Lung |
Raised in pregnancy Transiently elevated with cannabis use |
|
LDH |
Diagnosis and monitoring of germ cell tumours |
Elevated in cardiac disease and benign liver disease Elevated in some anaemias relating to non-malignant disease |
- Created on .
- Last updated on .